- Blog
- Anthem room correction vs dirac live
- Human design test
- Sonicwall netextender download windows 8
- Itunes this is spinal tap soundtrack
- Hrd genes
- Smart operation panel not working
- Mujhse dosti karoge movie watch
- Microsoft flight simulator x torrents
- How to play dbz supersonic warriors 2 players
- Bluesoleil 10 keygen
- Homekit ceiling fan control
- Change cursor color hots
- Faceshift studio 2015 1-02 retail
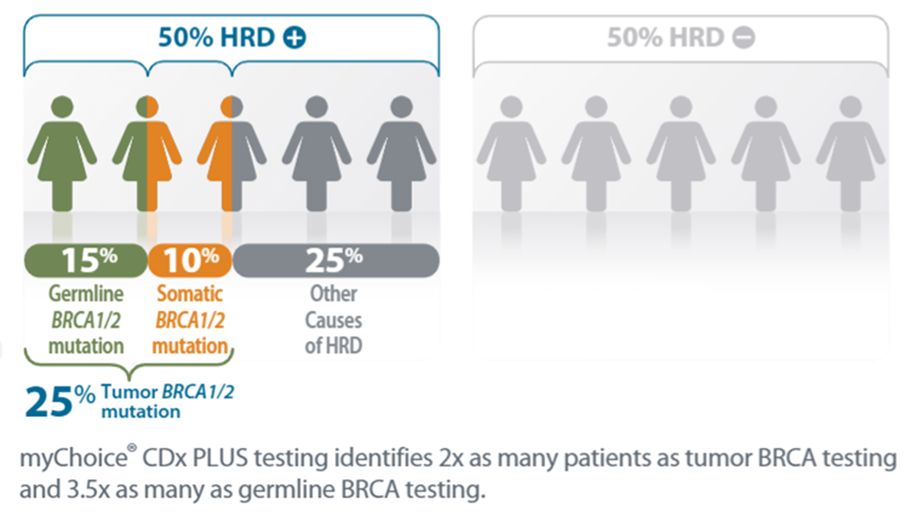

Fork degradation has been found to lead to chemosensitivity in BRCA-deficient tumors. Replication fork stalling can lead to fork degradation and decreased cell viability if forks do not resume DNA synthesis. DNA damage can also cause transient slowing or stalling of replication forks defined as replication stress. The inability to form RAD51 foci is a common feature of HRD. RAD51 foci formation has been used as a functional assay to define HRD and closely correlates with chemotherapy and PARPi sensitivity. Therefore, a functional HRD assay should be able to more accurately predict tumor response in real-time. However, these tests detect genomic scars that might not always correlate well with PARP inhibitor or platinum sensitivity in the current state. To date, methods for clinical detection of homologous recombination deficiency (HRD) are limited to genomic changes of HRR genes and genomic mutation patterns resulting from HRD genes involved in HR-mediated DNA repair. In tumors that have HRD, an alternate pathway such as non-homologous end-joining (NHEJ) is used and leads to error-prone repair. Thus, tumor cells with DSBs utilize the high-fidelity HRR as one of the central pathways for repair. Unrepaired DSBs are toxic to cells as genomic instability ensues and cells eventually die. Given platinum-based chemotherapy and poly (ADP-ribose) polymerase inhibitors (PARPis) are used in HGSOC, double-strand breaks (DSBs) are common. HRD can be caused by germline or somatic mutations of genes involved in the HR pathway. Approximately 50% of high-grade serous ovarian cancers (HGSOC) exhibit homologous recombination deficiency (HRD). Cancers with deficiencies in homologous recombination-mediated DNA repair (HRR) demonstrate improved clinical outcomes and increased survival.
- Blog
- Anthem room correction vs dirac live
- Human design test
- Sonicwall netextender download windows 8
- Itunes this is spinal tap soundtrack
- Hrd genes
- Smart operation panel not working
- Mujhse dosti karoge movie watch
- Microsoft flight simulator x torrents
- How to play dbz supersonic warriors 2 players
- Bluesoleil 10 keygen
- Homekit ceiling fan control
- Change cursor color hots
- Faceshift studio 2015 1-02 retail
